
Daridorexant, Approved for Adult Insomnia in the US, Canada, EU, Japan, Taiwan, and other marketsin 2026

Last updated: May 10, 2026
Quick Answer
Daridorexant, a prescription drug sold under the brand name QUVIVIQ, is an FDA-approved sleep medication for adults with insomnia. It works by blocking orexin receptors in the brain — chemicals that promote wakefulness — helping patients fall asleep faster, stay asleep longer, and feel better during the day. It is available in 25mg and 50mg doses taken once nightly.
Key Takeaways
- Daridorexant (QUVIVIQ) is a dual orexin receptor antagonist (DORA) approved for adult insomnia in the US, Canada, EU, Japan, Taiwan, and other markets.
- The 50mg dose significantly improves total sleep time, sleep onset, and wake after sleep onset compared to placebo, with effects confirmed up to one year [6].
- Side effects are minimal: adverse event rates in Phase 3 trials were comparable to placebo (13.41% vs. 14.81%) [2].
- Sales grew 74% year-on-year in Q1 2026, reflecting rapid adoption by physicians and patients [1].
- A pediatric Phase 2 trial showed statistically significant, dose-dependent sleep improvements in children, especially those with neurodevelopmental disorders [4].
- Daridorexant also reduces nocturia (nighttime urination) in older adults, adding a meaningful secondary benefit [5].
- Regulatory applications are expanding: South Korea filed in March 2026, with approval expected in 2027 [3].
- A label-expansion study is underway with the FDA to formally evaluate daytime functioning improvements [1].
What Is Daridorexant, a Prescription Drug?
Daridorexant, a prescription drug, is a dual orexin receptor antagonist (DORA) — a class of sleep medications that targets the brain’s wake-promoting system rather than broadly sedating the nervous system. It blocks both OX1R and OX2R orexin receptors, reducing the brain’s drive to stay awake at night.
Unlike older sleep aids such as benzodiazepines or Z-drugs, daridorexant does not cause widespread central nervous system depression. This makes it a more targeted option for chronic insomnia disorder in adults.
Brand name: QUVIVIQ
Available doses: 25mg and 50mg tablets
Dosing schedule: Once nightly, taken within 30 minutes of intended sleep time
Drug class: Dual orexin receptor antagonist (DORA)
How Does Daridorexant Work?
Daridorexant works by blocking orexin signaling — the brain’s primary system for maintaining wakefulness. When orexin is blocked at bedtime, the brain transitions more easily into sleep.
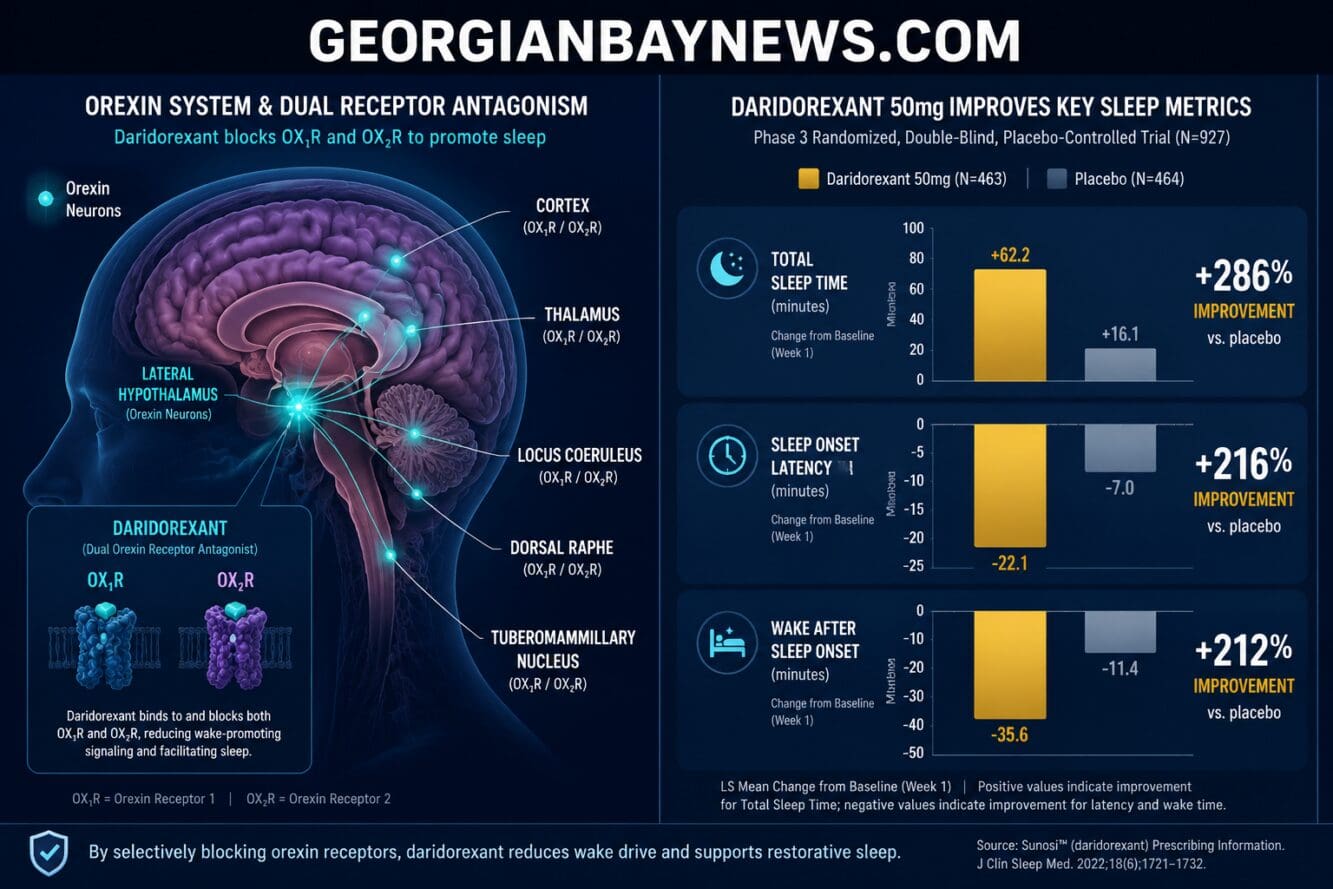
This mechanism differs from traditional sedatives in one key way: it doesn’t force sleep by suppressing overall brain activity. Instead, it removes a specific barrier to sleep. The result is a more natural sleep architecture, with patients retaining normal sleep stages including REM sleep.
Why this matters: Older sedatives like benzodiazepines can suppress deep sleep and carry significant dependency risks. Daridorexant’s targeted mechanism avoids both problems, making it a preferred option for long-term insomnia management [9].
What Does the Clinical Evidence Show for Daridorexant, a Prescription Drug?
The clinical data for daridorexant is strong across multiple sleep measures. In Phase 3 trials, the 50mg dose produced statistically significant improvements in:
| Sleep Measure | Improvement vs. Placebo | Significance |
|---|---|---|
| Subjective total sleep time (sTST) | +56.6 min vs. +35.7 min (placebo) at week 4 | p<0.0001 |
| Latency to sleep onset (sLSO) | Significantly reduced | p<0.0001 |
| Wake after sleep onset (sWASO) | Significantly reduced | p<0.0001 |
| Sleep depth | Improved at weeks 1–4 | Significant |
| Sleep quality | Improved at weeks 1–3 | Significant |
Data sourced from Phase 3 trials and published analyses [2][3][5].
Long-term data confirms that nightly use of daridorexant 50mg maintains these benefits for up to one year across both objective and patient-reported measures, including in older adults [6].
Who Is Daridorexant Prescribed For?
Daridorexant is approved for adults with chronic insomnia disorder — defined as difficulty falling or staying asleep at least three nights per week for at least three months.
Choose the 50mg dose if: sleep onset and maintenance are both problematic, and the patient has no contraindications to the higher dose.
Choose the 25mg dose if: the patient is older, takes other CNS-active medications, or has sensitivity concerns.
Daridorexant may not be appropriate for:
- Patients with narcolepsy (orexin deficiency is already present)
- Patients who cannot dedicate at least 7 hours to sleep
- Pregnant or breastfeeding individuals (consult a physician)
- Those taking strong CYP3A4 inhibitors (dose adjustment required)
What Are the Side Effects of Daridorexant, a Prescription Drug?
Daridorexant has a favorable safety profile. In the South Korean Phase 3 trial, treatment-emergent adverse events occurred in 13.41% of daridorexant patients versus 14.81% of placebo patients — essentially no difference [2].
Most commonly reported side effects include:
- Headache
- Somnolence (daytime drowsiness, generally mild)
- Dizziness
- Nausea
Rare but serious risks to discuss with a prescriber:
- Sleep paralysis
- Hypnagogic/hypnopompic hallucinations
- Complex sleep behaviors (sleepwalking, sleep-driving) — rare
Common mistake: Patients sometimes take daridorexant and then attempt to drive within a few hours. The prescribing information recommends allowing a full night of sleep (7+ hours) after taking the medication.
Does Daridorexant Help With Daytime Functioning?
Yes, and this is an area of active regulatory development. Beyond nighttime sleep, daridorexant significantly improved scores on the Insomnia Daytime Symptoms and Impacts Questionnaire (IDSIQ) compared to placebo [5].
Idorsia is currently conducting a label-enabling study in consultation with the US FDA to formally capture daytime functioning improvements on the drug’s label — a potential expansion beyond nighttime sleep metrics alone [1].
For adults aged 55 and older, daridorexant also reduced nocturia episodes (nighttime urination), with a median reduction of 31 minutes at week 1 (p=0.0027), adding a meaningful secondary quality-of-life benefit [5].
Where Is Daridorexant Currently Approved and Available?
As of 2026, daridorexant (QUVIVIQ) is approved or commercially available in:
- United States (FDA-approved; marketed by Idorsia)
- Canada (marketed by Idorsia)
- European Union (co-promotion expanding to additional markets) [2]
- Japan (marketed via Nxera/Shionogi partnership)
- Taiwan (approved in 25mg and 50mg formulations) [7]
- Hong Kong and China (marketed by Simcere)
- South Korea (MAA submitted March 2026; approval anticipated 2027) [3]
Sales grew 74% year-on-year in Q1 2026, indicating strong and accelerating global uptake [1].
Is Daridorexant Being Studied in Children?
Yes. Idorsia’s Phase 2 pediatric trial produced statistically significant, dose-dependent improvements in total sleep time in children with insomnia. Benefits were especially pronounced in children with co-morbid neurodevelopmental disorders such as ADHD or autism spectrum disorder [4].
Safety was confirmed even at the adult-recommended 50mg dose. Full data analysis and regulatory discussions with health authorities are ongoing, positioning daridorexant as a potential first-in-class treatment for pediatric insomnia [1][4].
Conclusion: What Should You Do Next?
Daridorexant, a prescription drug, represents a meaningful step forward in insomnia treatment. Its targeted mechanism, strong efficacy data, and minimal side effect profile make it a credible long-term option for adults struggling with chronic sleep problems.
Actionable next steps:
- Talk to your doctor if you have chronic insomnia lasting more than three months. Daridorexant requires a prescription and a proper clinical evaluation.
- Ask about the right dose — 25mg or 50mg — based on your age, other medications, and sleep patterns.
- Track your sleep before and after starting treatment using a sleep diary or wearable device to measure real-world improvement.
- Do not combine with alcohol or other CNS depressants without medical guidance.
- Follow global developments — regulatory approvals in South Korea and potential pediatric indications could expand access significantly by 2027.
For readers interested in broader health topics and community wellness coverage, explore our health and lifestyle content for additional context on managing wellbeing.
FAQ
Q: What is daridorexant used for?
Daridorexant is used to treat chronic insomnia disorder in adults. It helps people fall asleep faster, stay asleep longer, and feel more functional during the day.
Q: Is daridorexant a controlled substance?
In the United States, daridorexant is classified as a Schedule IV controlled substance, the same category as other sleep medications like zolpidem.
Q: How long does it take for daridorexant to work?
Clinical trials show meaningful improvements in sleep onset and total sleep time beginning in the first week of treatment, with continued benefit over months of use.
Q: Can older adults take daridorexant?
Yes. Clinical evidence confirms efficacy and tolerability in older adults, including a secondary benefit of reducing nighttime urination episodes [5].
Q: Is daridorexant better than zolpidem?
Daridorexant uses a more targeted mechanism with a comparable or better safety profile in trials. Direct head-to-head comparisons are limited, so discuss options with a prescriber.
Q: What is the brand name for daridorexant?
The brand name is QUVIVIQ.
Q: Can daridorexant be used in children?
Not yet approved for pediatric use, but Phase 2 results are promising. Regulatory discussions are ongoing as of 2026 [4].
Q: Does daridorexant cause next-day drowsiness?
Daytime somnolence was reported at low rates in trials, comparable to placebo. Allowing a full 7+ hours of sleep after taking the medication reduces this risk.
Q: How is daridorexant different from older sleep pills?
It blocks specific wake-promoting brain signals rather than broadly sedating the nervous system, which reduces risks of dependency and disrupted sleep architecture.
Q: Is daridorexant available in Canada?
Yes. Idorsia markets QUVIVIQ in Canada as of 2026.
References
[1] Media Release Details (Idorsia Q1 2026 Report) – https://www.idorsia.com/media/news/news-archive/media-release-details?id=3673709
[2] Nxera Positive Outcomes Daridorexant Study – https://www.clinicaltrialsarena.com/news/nxera-positive-outcomes-daridorexant-study/
[3] Nxera Pharma Submits Marketing Authorization Application For Daridorexant In South Korea – https://www.biospace.com/press-releases/nxera-pharma-submits-marketing-authorization-application-for-daridorexant-in-south-korea
[4] Pediatric Insomnia Phase 2 Results Presentation – https://www.idorsia.com/dam/jcr:dc383fbb-eb9a-4597-a7b6-9dd2e2f7a03a/260331_pediatric-insomnia-p2-results_presentation.pdf
[5] Daridorexant Improves Sleep Comorbid Nocturia With Insomnia – https://www.psychiatryadvisor.com/news/daridorexant-improves-sleep-comorbid-nocturia-with-insomnia/
[6] Long-Term Efficacy Review – https://www.tandfonline.com/doi/full/10.1080/14737175.2026.2639557
[7] Taiwan Approval – FirstWord Pharma – https://firstwordpharma.com/story/7180281
[9] Daridorexant Transforming Insomnia Management – https://medicalupdateonline.com/2026/01/daridorexant-transforming-insomnia-management/
Content, illustrations, and third-party video appearing on GEORGIANBAYNEWS.COM may be generated or curated with AI assistance or reproduced pursuant to the fair dealing provisions of the Copyright Act, R.S.C. 1985, c. C-42. Attribution and hyperlinks to original sources are provided in acknowledgment of applicable intellectual property rights. Such referencing is intended to direct traffic to and support the original rights holders’ platforms.






























