Canada just made pharmaceutical history. On April 28, 2026, Health Canada authorized the first generic semaglutide injection in any G7 nation—followed by a second approval just three days later [1][3]. For the estimated 12 million Canadians living with obesity or type 2 diabetes, this Health Canada Generic Ozempic Approval: Rollout Timeline, Price Drops, and Access Guide for Weight Loss Seekers represents a seismic shift in affordability and access to one of the most sought-after medications in the world.
The branded version of Ozempic has cost Canadians between $300 and $400+ per month out of pocket [2]. With multiple generics now entering the market, those prices could plummet to roughly $100 or less within months [7]. Here’s everything weight loss seekers need to know about eligibility, timelines, pricing, and safety.
Key Takeaways
- 🇨🇦 Canada is the first G7 country to approve generic semaglutide, with two products authorized in late April/early May 2026 [1][3].
- 💰 Prices could drop 50–65% once three or more generics reach pharmacy shelves, potentially bringing monthly costs below $100 CAD [7].
- 🏥 Pharmacy availability for Apotex’s generic is expected within weeks of the May 1, 2026 approval [2].
- 📋 Seven additional generic submissions are under Health Canada review, with decisions expected in coming weeks and months [1].
- ⚠️ Safety standards are identical to brand-name Ozempic—both approved generics met rigorous efficacy, safety, and quality criteria [3].
How the Health Canada Generic Ozempic Approval Happened So Quickly
The speed of these approvals surprised many observers. The Canadian patent for Ozempic expired in January 2026, immediately removing intellectual property barriers for generic manufacturers [7]. Health Canada completed its review of the Dr. Reddy’s Laboratories submission within the standard 180-day target timeline, demonstrating efficient regulatory processing [3].
Three days after that landmark first approval on April 28, 2026, Canadian-based Apotex received authorization for its product, marketed as Apo-Semaglutide Injection [1][8]. This rapid sequence signals Health Canada’s commitment to expanding medication access.
“Canada becomes the first G7 country to approve a generic version of semaglutide.” — Health Canada, April 28, 2026 [3]
Both products underwent comprehensive evidence review and met Health Canada’s rigorous standards for:
- Bioequivalence to brand-name Ozempic
- Safety profiles matching the original drug
- Quality manufacturing standards
- Efficacy for approved indications [3][1]
Rollout Timeline: When Can You Get Generic Semaglutide?
Understanding the rollout timeline is critical for weight loss seekers planning their next steps.
| Milestone | Date/Timeframe | Details |
|---|---|---|
| First generic approved (Dr. Reddy’s) | April 28, 2026 | Health Canada authorization [3] |
| Second generic approved (Apotex) | May 1, 2026 | Apo-Semaglutide Injection [1] |
| Apotex pharmacy availability | Summer 2026 (weeks after approval) | Confirmed by Apotex VP [2] |
| Seven additional generics under review | Coming weeks/months | Regulatory decisions pending [1] |
| Full market competition (3+ generics) | Late 2026 (estimated) | Maximum price reductions expected |
Catherine Thomas, Apotex’s vice president of global communications, confirmed the company’s generic should be available to Canadians within weeks of the May 1 approval [2]. Negotiations with private insurers regarding coverage are ongoing.
Price Drops: How Much Will You Save?
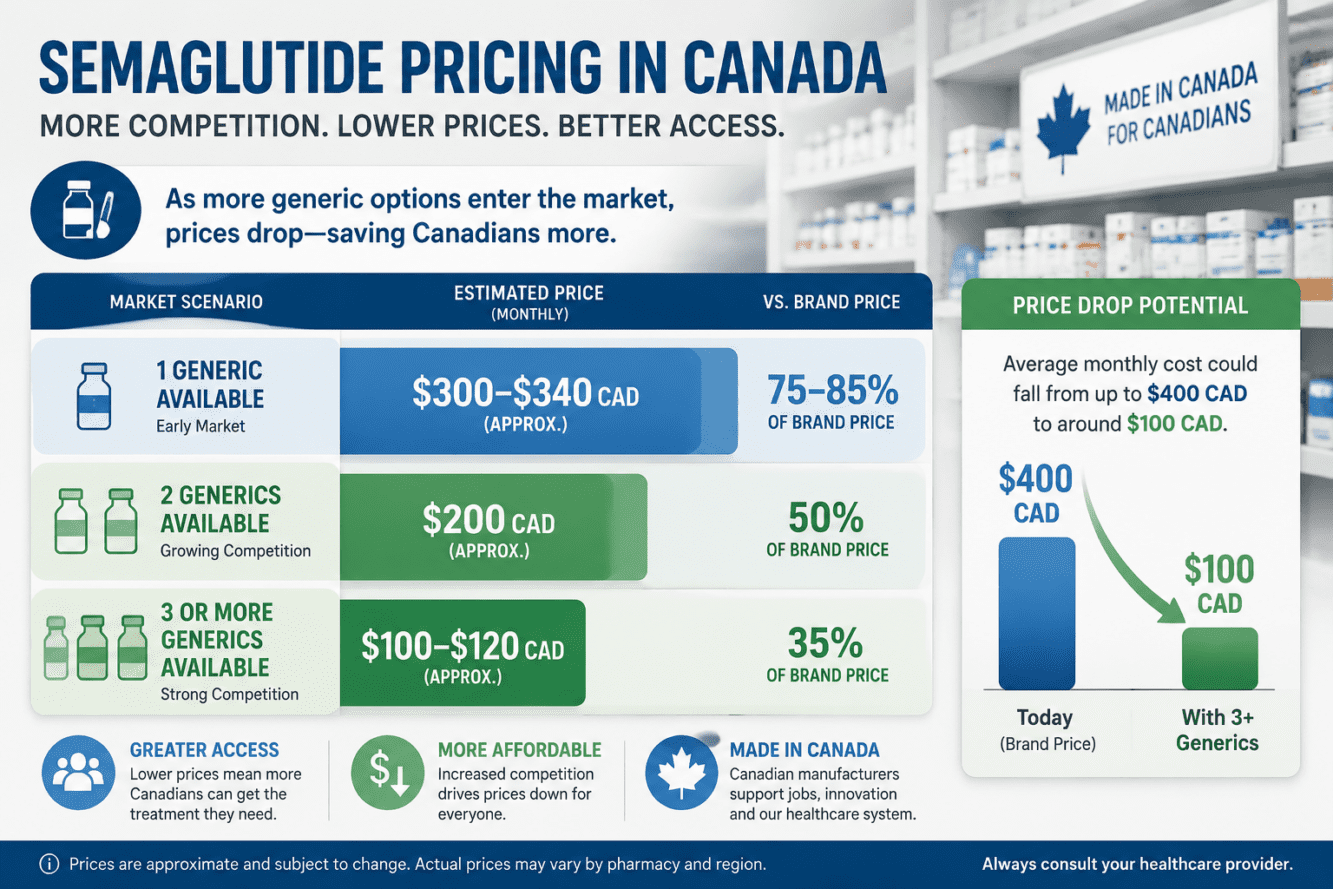
This is where the Health Canada Generic Ozempic Approval: Rollout Timeline, Price Drops, and Access Guide for Weight Loss Seekers delivers its most tangible impact. The pan-Canadian Pharmaceutical Alliance (pCPA) pricing framework establishes a tiered structure that rewards market competition [2]:
Tiered Pricing Breakdown
| Market Condition | Generic Price (% of Brand) | Estimated Monthly Cost |
|---|---|---|
| Brand-name only (previous) | 100% | $300–$663 CAD |
| One generic available | 75–85% of brand | ~$225–$340 CAD |
| Two generics available | 50% of brand | ~$150–$200 CAD |
| Three or more generics | ~35% of brand | ~$100 CAD or less |
With two generics already approved and seven more under review [1], the market is rapidly approaching the three-or-more threshold. Apotex has committed to offering Apo-Semaglutide at “a fraction of the cost” of brand-name Ozempic, following pCPA pricing guidelines [8].
Current brand-name pricing context: Canadian pharmacy listings show Ozempic ranging from $222 to $663 CAD depending on dosage and pharmacy location [2]. Even at the 50% tier, savings of $150–$300 per month are realistic for most patients.
Access Guide: Eligibility, Prescriptions, and Insurance Coverage
Who Can Get Generic Semaglutide?
Generic semaglutide carries the same approved indications as brand-name Ozempic. In Canada, semaglutide is currently approved for:
- Type 2 diabetes management (primary indication)
- Chronic weight management in adults with obesity (BMI ≥30) or overweight (BMI ≥27) with at least one weight-related comorbidity
A valid prescription from a licensed healthcare provider is required. Patients should discuss with their doctor whether switching from brand-name to generic is appropriate.
Steps to Access Generic Semaglutide
- Consult your physician — Confirm eligibility based on BMI, health history, and treatment goals
- Request the generic — Ask your prescriber to write for generic semaglutide or allow substitution
- Check pharmacy availability — Call ahead; supply may be limited in early weeks
- Contact your insurer — Verify whether your plan covers generic semaglutide (negotiations are ongoing) [2]
- Explore provincial formularies — Coverage decisions vary by province
Insurance and Coverage Considerations
Private insurers are currently negotiating coverage terms for generic semaglutide [2]. Patients should:
- ✅ Contact their benefits provider directly
- ✅ Ask about prior authorization requirements
- ✅ Inquire about step therapy protocols
- ✅ Check if their plan distinguishes between diabetes and weight management indications
Side Effects and Safety: What Weight Loss Seekers Should Know
Generic semaglutide has the identical active ingredient as brand-name Ozempic. This means the side effect profile is the same [3][1].
Common Side Effects
- Nausea (especially during dose escalation)
- Vomiting
- Diarrhea
- Constipation
- Abdominal pain
- Decreased appetite
Serious Side Effects (Rare)
- Pancreatitis
- Gallbladder problems
- Kidney injury
- Thyroid tumors (observed in animal studies)
- Severe allergic reactions
Health Canada confirmed it will continue to supervise the safety and efficacy of all semaglutide products through post-approval surveillance [7]. Patients experiencing unusual symptoms should contact their healthcare provider immediately.
⚠️ Important: Semaglutide is not appropriate for everyone. People with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 should not use this medication.
How Generic Semaglutide Disrupts the GLP-1 Market for Obesity Treatment
The approval of generic semaglutide in Canada sends ripple effects across the global GLP-1 receptor agonist market. Key disruptions include:
- Reduced financial barriers — Millions of Canadians previously priced out can now consider treatment
- Pressure on brand-name pricing — Novo Nordisk faces competitive pressure to adjust Ozempic pricing
- Precedent for other nations — Other G7 countries may accelerate their own generic pathways
- Expanded treatment population — Lower costs mean more physicians may recommend semaglutide for eligible patients
- Supply chain diversification — Multiple manufacturers reduce shortage risks that plagued Ozempic in 2023–2024
With seven additional generics in the regulatory pipeline [1], Canada’s semaglutide market is poised for robust competition by late 2026.
Conclusion
The Health Canada Generic Ozempic Approval: Rollout Timeline, Price Drops, and Access Guide for Weight Loss Seekers marks a turning point for medication affordability in Canada. Two generics are approved, seven more are under review, and pharmacy availability is expected by summer 2026.
Actionable next steps for weight loss seekers:
- 📞 Book an appointment with your healthcare provider to discuss semaglutide eligibility
- 💊 Ask about generic substitution if you’re already on brand-name Ozempic
- 📋 Contact your insurer to understand upcoming coverage changes
- ⏰ Monitor pharmacy availability — Apotex’s product should arrive within weeks [2]
- 🔍 Stay informed — Additional approvals will trigger further price drops
With monthly costs potentially falling below $100 CAD once three or more generics reach the market [7], effective weight management medication is becoming accessible to far more Canadians than ever before.
References
[1] Canada Approves Second Generic Semaglutide The First G7 Country To Do So – https://www.canada.ca/en/health-canada/news/2026/05/canada-approves-second-generic-semaglutide-the-first-g7-country-to-do-so.html
[2] Experts Welcome Generic Semaglutide Urge Caution Reliance May 2026 – https://glp1prices.ca/glp1-news/experts-welcome-generic-semaglutide-urge-caution-reliance-may-2026
[3] Canada Becomes The First G7 Country To Approve A Generic Version Of Semaglutide – https://www.canada.ca/en/health-canada/news/2026/04/canada-becomes-the-first-g7-country-to-approve-a-generic-version-of-semaglutide.html
[7] Ozempic Generic Health Canada 9 – https://www.cbc.ca/news/health/ozempic-generic-health-canada-9.7180566
[8] Cp Newsalert Health Canada Approves 2nd Generic Version Of Ozempic And Its Canadian – https://www.cjme.com/2026/05/01/cp-newsalert-health-canada-approves-2nd-generic-version-of-ozempic-and-its-canadian/












